In July 2018, FDA released the Biosimilars Action Plan (BAP), which outlined FDA’s approach for expanding access to biosimilars for the American public. The plan focused on 4 key areas:
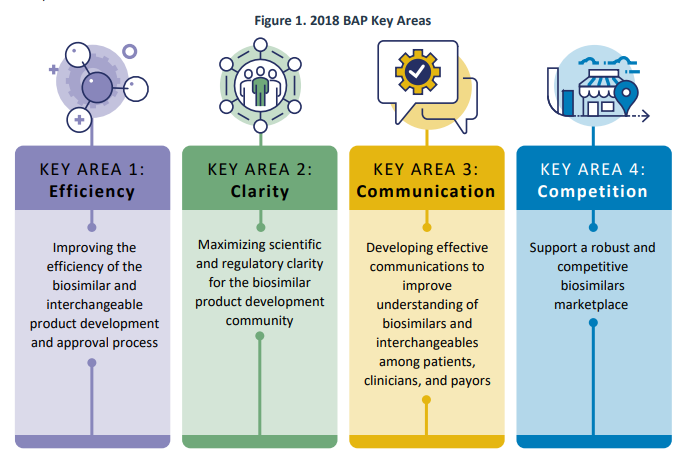
A recent FDA report reviews some of their accomplishments since then. Most of these efforts revolve around guidance documents, additional staff, education products and websites, public hearings and regulations (i.e., proposed/final rules). FDA also added new data resources including publishing a modernized version of the Purple Book in February 2020. FDA also collaborated with other agencies such as FTC, and produced a joint statement and held a workshop in March 2020, entitled: “Public Workshop: FDA/FTC Workshop on a Competitive Marketplace for Biosimilars.” Some other key actions are listed below.
The full report is here.












